No edit summary |
|||
| (122 intermediate revisions by 6 users not shown) | |||
| Line 1: | Line 1: | ||
[[Category:benchmark]] | [[Category:benchmark]] | ||
[[Category: | [[Category:PDE]] | ||
[[Category: | [[Category:nonlinear]] | ||
[[Category:Parametric]] | |||
[[Category: | |||
==Description== | |||
<math> | <figure id="fig:bach">[[File:Fig_BatchChrom.jpg|490px|thumb|right|<caption>Sketch of a batch chromatographic process for the separation of A and B.</caption>]]</figure> | ||
\frac{\partial c_i}{\partial t}+\frac{1-\epsilon}{\epsilon}\frac{\partial q_i}{\partial t}+u\frac{\partial c_i}{\partial z}-D_i\frac{\partial^2 c_i}{\partial z^2}=0, \qquad z\in(0, | |||
Preparative liquid chromatography as a crucial separation and purification tool has been widely employed in the food, fine chemical, and pharmaceutical industries. Chromatographic separation at the industry scale can be operated either discontinuously or in a continuous mode. | |||
The continuous case will be discussed in the benchmark [[SMB]], and here we focus on the discontinuous mode -- [[wikipedia:Affinity_chromatography#Batch_and_column_setups|batch chromatography]]. | |||
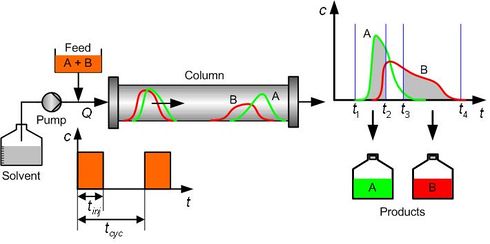
The principle of the batch chromatographic process for the binary separation is shown in Fig. 1. | |||
During the injection period <math>t_{inj}</math>, a mixture of products A and B is injected at the inlet of the column packed with a suitable stationary phase. | |||
With the help of the mobile phase, the feed mixture flows through the column. | |||
Since the to-be-separated solutes exhibit different adsorption affinities to the stationary phase, they move at different velocities | |||
and thus separate from each other when exiting the column. | |||
At the column outlet, component A is collected between <math>t_1</math> and <math>t_2</math>, and component B is collected between <math>t_3</math> and <math>t_4</math>. | |||
Here the positions of <math>t_1</math> and <math>t_4</math> are determined by a minimum concentration threshold that the detector can resolve, and the positions of <math>t_2</math> and <math>t_3</math> are determined by the purity specifications imposed on the products. | |||
After the cycle period <math>t_{cyc}:=t_4-t_1</math>, the injection is repeated. | |||
The feed flow rate <math>Q</math> and injection period <math>t_{inj}</math> are often considered as the operating variables. | |||
By properly choosing them, the process can achieve the desired performance criterion, such as production rate, while respecting the product specifications (e.g., purity, recovery yield). | |||
The dynamics of the batch chromatographic column can be described precisely by an axially dispersed plug-flow model with a limited mass-transfer rate characterized by a linear driving force (LDF) approximation. | |||
In this model the differential mass balance of component <math>i</math> (<math>i=A,B,</math>) in the liquid phase can be written as: | |||
:<math> | |||
\frac{\partial c_i}{\partial t} | |||
+ \frac{1 - \epsilon}{\epsilon} \frac{\partial q_i}{\partial t} | |||
+ u \frac{\partial c_i}{\partial z} | |||
- D_i \frac{\partial^2 c_i}{\partial z^2} | |||
= 0, \qquad | |||
z \in (0, L), \qquad (1) | |||
</math> | </math> | ||
where <math>c_i</math> and <math>q_i</math> are the concentrations of solute <math>i</math> in the liquid and solid phases, respectively, <math>u</math> the interstitial liquid velocity, <math>\epsilon</math> the column porosity, <math>t</math> the time coordinate, <math>z</math> the axial coordinate along the column, <math>L</math> the column length, <math>D_i=\frac{uL}{Pe}</math> the axial dispersion coefficient and <math>Pe</math> the Péclet number. The adsorption rate is modeled by the LDF approximation: | where <math>c_i</math> and <math>q_i</math> are the concentrations of solute <math>i</math> in the liquid and solid phases, respectively, <math>u</math> the interstitial liquid velocity, <math>\epsilon</math> the column porosity, <math>t</math> the time coordinate, <math>z</math> the axial coordinate along the column, <math>L</math> the column length, <math>D_i=\frac{uL}{Pe}</math> the axial dispersion coefficient and <math>Pe</math> the [[wikipedia:Péclet_number|Péclet number]]. | ||
The adsorption rate is modeled by the LDF approximation: | |||
<math> | :<math> | ||
\frac{\partial q_i}{\partial t} = | \frac{\partial q_i}{\partial t} | ||
= \kappa_{i} \left(q^{Eq}_i - q_i\right), \qquad | |||
z \in [0, L], | |||
</math> | </math> | ||
where <math> | where <math>\kappa_{i}</math> is the mass-transfer coefficient of component <math>i</math> and <math>q^{Eq}_i</math> is the adsorption equilibrium concentration calculated by the isotherm equation for component <math>i</math>. Here the bi-[[wikipedia:Langmuir_adsorption_model|Langmuir]] isotherm model is used to describe the adsorption equilibrium: | ||
<math> | :<math> | ||
q^{Eq}_i=\frac{H_{i,1}\,c_i}{1+\sum_{j=A,B}K_{j,1}\,c_j}+\frac{H_{i,2}\,c_i}{1+\sum_{j=A,B}K_{j,2}\,c_j},\; i=A,B, | q^{Eq}_i | ||
= \frac{H_{i,1}\,c_i}{1 + \sum_{j = A, B}K_{j,1}\,c_j} | |||
+ \frac{H_{i,2}\,c_i}{1 + \sum_{j = A, B}K_{j,2}\,c_j},\; | |||
i = A, B, | |||
</math> | </math> | ||
where <math>H_{i,1}</math> and <math>H_{i,2}</math> are the Henry constants, and <math>K_{j,1}</math> and <math>K_{j,2}</math> the thermodynamic coefficients. | where <math>H_{i,1}</math> and <math>H_{i,2}</math> are the Henry constants, and <math>K_{j,1}</math> and <math>K_{j,2}</math> the thermodynamic coefficients. | ||
The boundary conditions for | The boundary conditions for (1) are specified by the Danckwerts relations: | ||
<math> | :<math> | ||
D_i\left.\frac{\partial c_i}{\partial z}\right|_{z=0} = u\ | D_i \left.\frac{\partial c_i}{\partial z}\right|_{z = 0} | ||
= u \left(\left.c_i\right|_{z=0} - c^{in}_i\right), \qquad | |||
\left.\frac{\partial c_i}{\partial z}\right|_{z = L} | |||
= 0, | |||
</math> | </math> | ||
where <math>c^{in}_i</math> is the concentration of component <math>i</math> at the inlet of the column. A rectangular injection is assumed for the system and thus | where <math>c^{in}_i</math> is the concentration of component <math>i</math> at the inlet of the column. | ||
A rectangular injection is assumed for the system and thus | |||
\ | :<math> | ||
\ | c^{in}_i | ||
= | |||
\begin{cases} | |||
c^F_i, & \text{if } t \le t_{inj}, \\ | |||
0, & \text{if } t > t_{inj}. | |||
\end{cases} | |||
</math> | </math> | ||
Here <math>c^F_i</math> is the feed concentration for component <math>i</math> and <math>t_{inj}</math> is the injection period. | |||
In addition, the column is assumed unloaded initially: | |||
<math> | :<math> | ||
c_i(t=0,z)=q_i(t=0,z)=0,\quad z\in[0, | c_i(t = 0, z) | ||
= q_i(t = 0,z) | |||
= 0, \quad | |||
z \in [0, L], \; | |||
i = A, B. | |||
</math> | </math> | ||
More details about the mathematical modeling for batch chromatography can be found in the literature <ref name="guiochon06"/>. | |||
==Discretization== | ==Discretization== | ||
In this model, the feed flow-rate <math>Q</math> and injection period <math>t_{inj}</math> are | In this model, the feed volumetric flow-rate <math>Q</math> and the injection period <math>t_{inj}</math> are considered as the operating parameters, and denoted as the parameter <math>\mu=(Q,\,t_{inj})</math>. Using the finite volume discretization, we get the full order model (FOM) as follows, | ||
<math> | :<math> | ||
\left\{ | \left\{ | ||
\begin{ | \begin{aligned} | ||
\mathbf{ | \mathbf{A} \mathbf{c}_i^{k+1} | ||
- \frac{1-\epsilon}{\epsilon} \Delta t \mathbf | &= \mathbf{B} \mathbf{c}_i^{k} | ||
\mathbf{q}_i^{k+1} = \mathbf{q}_i^{k} + \Delta t \mathbf | + d_i^k | ||
\end{ | - \frac{1 - \epsilon}{\epsilon} \Delta t \mathbf{h}_i^k, \\ | ||
\right . | \mathbf{q}_i^{k + 1} | ||
&= \mathbf{q}_i^{k} | |||
+ \Delta t \mathbf{h}_i^k, | |||
\end{aligned} | |||
\right. | |||
</math> | </math> | ||
where <math>\mathbf{c}_i^k, \mathbf{q}_i^k \in \mathbb{R}^{\mathcal N}, i = A, B</math> are the solution vectors of <math>c_i</math> and <math>q_i</math> at the time instance <math>t = t^k, k = 0, 1, \ldots, K</math>, respectively. | |||
<math>k | The time step <math>\Delta t </math> is determined by the stability condition. | ||
determined by the stability condition. | The equation <math> \mathbf h_i^k = \kappa_{i} (\mathbf q^{Eq}_i - \mathbf q_i^k) </math>, is time- and parameter-dependent, the boldface <math>\mathbf{A,B}</math> are constant matrices. As a result, it is a nonlinear parametric system. | ||
==Generation of ROM== | ==Generation of ROM== | ||
For parametrized time-dependent problems, the reduced basis | The reduced order model (ROM) can be obtained by the reduced basis method <ref name="zhang15"/>, which is applicable for nonlinear parametric systems, see | ||
[[Reduced_Basis_PMOR_method|Reduced Basis PMOR method]]. | |||
For parametrized time-dependent problems, the reduced basis is often generated by using the POD-Greedy algorithm <ref name="haasdonk08"/>. | |||
<math> | Notice that the nonlinear functions <math>\mathbf h_i, i=A, B</math> can be approximated by the empirical interpolation method <ref name="barrault04"/>, | ||
such that the ROM can be obtained efficiently by the strategy of offline-online decomposition. | |||
Assume <math>W_z</math> is the collateral reduced basis (CRB) for the nonlinear operator <math>h_z</math>, and <math>V_{c_z},V_{q_z}</math> are the reduced bases for the field variables <math>c_z</math> and <math>q_z</math>, respectively. | |||
Applying Galerkin projection and empirical operator interpolation, the ROM can be formulated as: | |||
:<math> | |||
\left\{ | |||
\begin{aligned} | |||
\hat{A}_{c_z} {a}_{c_z}^{n+1} | |||
&= \hat{B}_{c_z} {a}_{c_z}^{n} | |||
+ d_0^n \hat{d}_{c_z} | |||
- \frac{1 - \epsilon}{\epsilon} \Delta t \hat{H}_{c_z} \beta_z^n, \\ | |||
{a}_{q_z}^{n + 1} | |||
&= {a}_{q_z}^{n} | |||
+ \Delta t \hat{H}_{q_z} \beta_z^n, | |||
\end{aligned} | |||
\right. | |||
</math> | |||
where <math> {a}_{c_z}^n, {a}_{q_z}^n \in \mathbb R^N</math> are the solution of the ROM. <math>\hat{A}_{c_z}=V_{c_z}^T A V_{c_z}, | |||
\hat{ B}_{c_z}=V_{c_z}^T B V_{c_z}, | |||
\hat{d}_{c_z}^{n}=V_{c_z}^T e_1</math>, | |||
<math>\hat{H}_{c_z}:= V_{c_z}^TW_z ,</math> | |||
<math>\hat{H}_{q_z}:= V_{q_z}^TW_z </math> are the reduced matrices, <math>e_1:=(1,0,\cdots,0)</math>. | |||
<math>\beta_z^n \in \mathbb R^M</math> is the coefficients of the CRB <math>W_z</math> for the empirical interpolation. | |||
==Data== | |||
<figure id="fig:cabfr">[[File:cabfrA.png|480px|thumb|right|<caption>Concentrations at the outlet of the column.</caption>]]</figure> | |||
Fig. 2 shows the concentrations at the outlet of the column at a given parameter <math>\mu= (0.1018, 1.3487)</math>, which show that the ROM (<math>N=46, M=151</math>) reproduces the dynamics of the full order model (<math>\mathcal N=1000</math>). | |||
==Citation== | |||
To cite this benchmark, use the following references: | |||
* For the benchmark itself and its data: | |||
::The MORwiki Community, '''Batch Chromatography'''. MORwiki - Model Order Reduction Wiki, 2018. http://modelreduction.org/index.php/Batch_Chromatography | |||
@MISC{morwiki_bone, | |||
author = <nowiki>{{The MORwiki Community}}</nowiki>, | |||
title = {Batch Chromatography}, | |||
howpublished = {{MORwiki} -- Model Order Reduction Wiki}, | |||
url = <nowiki>{https://modelreduction.org/morwiki/index.php/Batch_Chromatography}</nowiki>, | |||
year = {2018} | |||
} | |||
* For the background on the benchmark: | |||
@ARTICLE{RieWHetal95, | |||
author = <nowiki>{Y. Zhang and L. Feng and S. Li and P. Benner}</nowiki>, | |||
title = {Accelerating {PDE} constrained optimization by the reduced basis method: application to batch chromatography}, | |||
journal = {International Journal for Numerical Methods in Engineering}, | |||
volume = {104}, | |||
number = {11}, | |||
pages = {983--1007}, | |||
year = {2015}, | |||
doi = {10.1002/nme.4950} | |||
} | |||
==References== | |||
<references> | |||
<ref name="guiochon06"> G. Guiochon, A. Felinger, D. G. Shirazi, A. M. Katti, <span class="plainlinks">[http://books.google.com/books/about/Fundamentals_of_preparative_and_nonlinea.html?id=UjZRAAAAMAAJ Fundamentals of Preparative and Nonlinear Chromatography]</span>, 2nd Edition, Academic Press, 2006.</ref> | |||
<ref name="barrault04"> M. Barrault, Y. Maday, N.C. Nguyen, and A.T. Patera, "<span class="plainlinks">[https://doi.org/10.1016/j.crma.2004.08.006 An 'empirical interpolation' method: application to efficient reduced-basis discretization of partial differential equations]</span>", C. R. Acad. Sci. Paris Series I, 339 (2004), 667-672.</ref> | |||
== | <ref name="haasdonk08"> B. Haasdonk and M. Ohlberger, "<span class="plainlinks">[https://doi.org/10.1051/m2an:2008001 Reduced basis method for finite volume approximations of parameterized linear evolution equations]</span>", Mathematical Modeling and Numerical Analysis, 42 (2008), 277-302.</ref> | ||
<ref name="zhang15"> Y. Zhang, L. Feng, S. Li and P. Benner, "<span class="plainlinks">[https://doi.org/10.1002/nme.4950 Accelerating PDE constrained optimization by the reduced basis method: application to batch chromatography]</span>", International Journal for Numerical Methods in Engineering, 104(11): 983--1007, 2015.</ref> | |||
Contact | </references> | ||
==Contact== | |||
'' [[User:Zhangy|Yongjin Zhang]]'' | '' [[User:Zhangy|Yongjin Zhang]]'' | ||
'' [[User:Suzhou|Suzhou Li]]'' | |||
Latest revision as of 08:59, 11 June 2025
Description
Preparative liquid chromatography as a crucial separation and purification tool has been widely employed in the food, fine chemical, and pharmaceutical industries. Chromatographic separation at the industry scale can be operated either discontinuously or in a continuous mode. The continuous case will be discussed in the benchmark SMB, and here we focus on the discontinuous mode -- batch chromatography. The principle of the batch chromatographic process for the binary separation is shown in Fig. 1. During the injection period , a mixture of products A and B is injected at the inlet of the column packed with a suitable stationary phase. With the help of the mobile phase, the feed mixture flows through the column. Since the to-be-separated solutes exhibit different adsorption affinities to the stationary phase, they move at different velocities and thus separate from each other when exiting the column. At the column outlet, component A is collected between and , and component B is collected between and . Here the positions of and are determined by a minimum concentration threshold that the detector can resolve, and the positions of and are determined by the purity specifications imposed on the products. After the cycle period , the injection is repeated. The feed flow rate and injection period are often considered as the operating variables. By properly choosing them, the process can achieve the desired performance criterion, such as production rate, while respecting the product specifications (e.g., purity, recovery yield).
The dynamics of the batch chromatographic column can be described precisely by an axially dispersed plug-flow model with a limited mass-transfer rate characterized by a linear driving force (LDF) approximation. In this model the differential mass balance of component () in the liquid phase can be written as:
where and are the concentrations of solute in the liquid and solid phases, respectively, the interstitial liquid velocity, the column porosity, the time coordinate, the axial coordinate along the column, the column length, the axial dispersion coefficient and the Péclet number. The adsorption rate is modeled by the LDF approximation:
where is the mass-transfer coefficient of component and is the adsorption equilibrium concentration calculated by the isotherm equation for component . Here the bi-Langmuir isotherm model is used to describe the adsorption equilibrium:
where and are the Henry constants, and and the thermodynamic coefficients.
The boundary conditions for (1) are specified by the Danckwerts relations:
where is the concentration of component at the inlet of the column. A rectangular injection is assumed for the system and thus
Here is the feed concentration for component and is the injection period. In addition, the column is assumed unloaded initially:
More details about the mathematical modeling for batch chromatography can be found in the literature [1].
Discretization
In this model, the feed volumetric flow-rate and the injection period are considered as the operating parameters, and denoted as the parameter . Using the finite volume discretization, we get the full order model (FOM) as follows,
where are the solution vectors of and at the time instance , respectively. The time step is determined by the stability condition. The equation , is time- and parameter-dependent, the boldface are constant matrices. As a result, it is a nonlinear parametric system.
Generation of ROM
The reduced order model (ROM) can be obtained by the reduced basis method [2], which is applicable for nonlinear parametric systems, see Reduced Basis PMOR method. For parametrized time-dependent problems, the reduced basis is often generated by using the POD-Greedy algorithm [3]. Notice that the nonlinear functions can be approximated by the empirical interpolation method [4], such that the ROM can be obtained efficiently by the strategy of offline-online decomposition.
Assume is the collateral reduced basis (CRB) for the nonlinear operator , and are the reduced bases for the field variables and , respectively. Applying Galerkin projection and empirical operator interpolation, the ROM can be formulated as:
where are the solution of the ROM. , are the reduced matrices, . is the coefficients of the CRB for the empirical interpolation.
Data
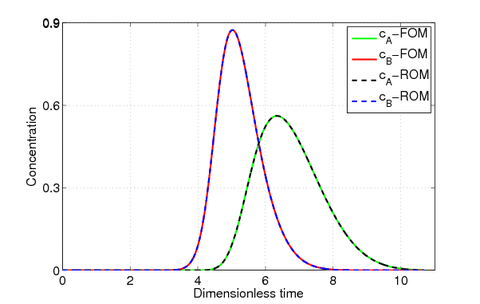
Fig. 2 shows the concentrations at the outlet of the column at a given parameter , which show that the ROM () reproduces the dynamics of the full order model ().
Citation
To cite this benchmark, use the following references:
- For the benchmark itself and its data:
- The MORwiki Community, Batch Chromatography. MORwiki - Model Order Reduction Wiki, 2018. http://modelreduction.org/index.php/Batch_Chromatography
@MISC{morwiki_bone,
author = {{The MORwiki Community}},
title = {Batch Chromatography},
howpublished = {{MORwiki} -- Model Order Reduction Wiki},
url = {https://modelreduction.org/morwiki/index.php/Batch_Chromatography},
year = {2018}
}
- For the background on the benchmark:
@ARTICLE{RieWHetal95,
author = {Y. Zhang and L. Feng and S. Li and P. Benner},
title = {Accelerating {PDE} constrained optimization by the reduced basis method: application to batch chromatography},
journal = {International Journal for Numerical Methods in Engineering},
volume = {104},
number = {11},
pages = {983--1007},
year = {2015},
doi = {10.1002/nme.4950}
}
References
- ↑ G. Guiochon, A. Felinger, D. G. Shirazi, A. M. Katti, Fundamentals of Preparative and Nonlinear Chromatography, 2nd Edition, Academic Press, 2006.
- ↑ Y. Zhang, L. Feng, S. Li and P. Benner, "Accelerating PDE constrained optimization by the reduced basis method: application to batch chromatography", International Journal for Numerical Methods in Engineering, 104(11): 983--1007, 2015.
- ↑ B. Haasdonk and M. Ohlberger, "Reduced basis method for finite volume approximations of parameterized linear evolution equations", Mathematical Modeling and Numerical Analysis, 42 (2008), 277-302.
- ↑ M. Barrault, Y. Maday, N.C. Nguyen, and A.T. Patera, "An 'empirical interpolation' method: application to efficient reduced-basis discretization of partial differential equations", C. R. Acad. Sci. Paris Series I, 339 (2004), 667-672.